The new EU Medical Devices and In Vitro Diagnostical Medical Devices regulations are presenting huge challenges for medical device manufacturers, with thousands of products impacted and requiring label and IFU updates in order to be compliant. Additional content needs to be included on device packaging and labeling, including a Unique Device Identifier (UDI), new symbols and warning statements. Without an end-to-end artwork and labeling management system, manufacturers will find it almost impossible to comply.
How does Kallik help with EU MDR and IVDR?
|
|
Challenge |
How Kallik can help |
IDENTIFY & LOCATE |
Labels and IFUs are not often held in a central repository, meaning that it can be difficult to locate and identify these for update. Identifying which labels and IFUs require change is a manual process which can take weeks if not months to complete |
All labels and IFUs are stored in a central Asset Manager and impacted labels and IFUs are instantly identified using Veraciti™'s ‘Where Used’ functionality |
UPDATE |
Updating thousands of IFUs and labels is a resource-heavy activity and can take months to complete |
Mass changes can be made to thousands of artworks in hours, not months, using Veraciti™’s automated artwork capabilities |
CONTROL & COMPLY |
Managing the approval process using manual spreadsheet tracking and emails is risky, prone to error and makes proving compliance difficult |
Ensure compliance with automated approval routing, task management, e-signatures and detailed audit trails |
|
|
Label and IFU artworks need to be emailed to the factory or transferred into a separate system used for printing, creating a risk the wrong version is printed |
Easily print the finished labels directly from the same system, ensuring only the latest version can be printed |
DISTRIBUTE |
Updated artwork files are emailed or transferred to third party printers using manual processes, risking the wrong version of the label being printed. eIFUs need to be manually uploaded to the relevant platform, risking misalignment between the approved file and the one published online. |
Instantly distribute the approved artwork to third party printers from within the system. eIFUs are instantly published following approval, meaning the latest version is always accessible to the patient. |
Medical Device Labels Before VS After MDR

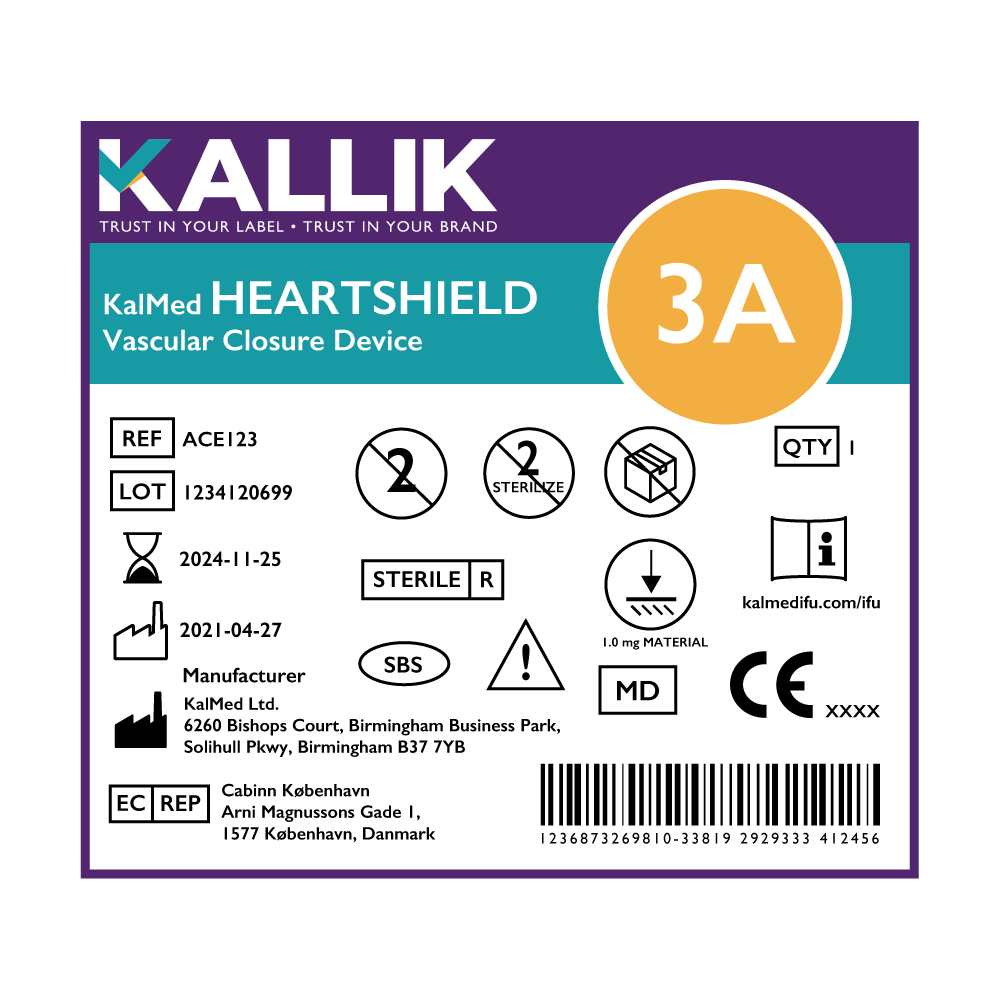
Talk to the experts in compliant labeling and artwork
Kallik’s labeling and artwork management software, Veraciti, has been specifically developed for the needs of medical device manufacturers. We have supported many medical device companies with their journey towards EU MDR and IVDR compliant labeling and artwork. Our experienced team can help you, too.
Check out our latest Medical Device resources
Why New Medical Device Regulations Require More Than Ad-Hoc Manual Processes

This time around, products really do have to ‘do what they say on the tin’!
Reducing Errors In Factory Print: The Case For Labeling Digitalization

It is too risky to leave the accuracy of market-facing product information to chance
UDI & Product Traceability For Medical Devices: White Paper

How a more strategic approach to labeling digitalization can better serve businesses & regulators
Lessons & Leftover Challenges from MDR Preparations: What Next?

How medical device companies can effectively overcome the challenges posed by EU MDR
EU MDR Labeling Compliance - Are We There Yet?

Bob Tilling, VP Global Sales, explains how medical device manufacturers can recover lost time
How Companies Are Using New EU MDR Regulations To Improve Artwork & Labeling Processes

EU MDR forced changes on medical device manufacturers but has led to improvements in multiple areas
Key EU MDR Changes You Need To Know About

New medical device regulations substantially change the ways manufacturers market devices in the EU
What We Can Learn From EU Medical Device Regulation Compliance Preparations

MDR requirements alone should not determine how manufacturers overhaul their label management
Is Your Medical Device Organization On Track To Comply With EU MDR?

The clock is ticking on the need to ensure compliance with the EU Medical Device Regulation (EU MDR)
EU MDR: An Opportunity For Medical Device Manufacturers

Compliance is not a short-term business overhead, it is an opportunity to improve best practice
Labeling and the Art of Data Quality for UDI
How adopting best practice for managing labeling content can bring business value to UDI projects
Why UDI Is Europe's Latest Hot Topic

Not only does data have value, but structured data has increased value
How To Improve Return On Investment From UDI

The transition to UDI is a huge undertaking, so it is important companies take a wider perspective
Factory Printing: How To Reduce Errors, Accelerate Printing Times and Maintain Compliance

The risks of using manual processes for factory printing and how to successfully tackle them
Factory Labeling Migration Following Business Acquisition

Veraciti, with 21 CFR Part 11 electronic signatures, eliminated paper-based quality processes
Veraciti™ Sets New Quality Benchmarks at Integra LifeSciences

Surgical and medical instrument manufacturer Integra LifeSciences is a leader in medical technology
3 MDR Lessons Class II Should Learn From Class III Medical Device Businesses

Why it is vital medical device manufacturers begin planning a technology-led, long-term solution?
6 Steps Medical Device Manufacturers Must Take When Navigating The Digital Maturity Curve

For medical device manufacturers faced with tight regulations, manual processes are not feasible
Helping Device Manufacturers Get Those Vital Ventilators Built

Delivering ventilators to the NHS on time with required labeling during the peak of the pandemic
How To Ensure Full IVDR Compliance On Time

A guide on how your company can get compliant with new medical device regulations
4 Key Actions To Ensure IVDR Compliance Deadlines Are Met

The next challenge after EU MDR is IVDR. Don't let your business fall short of compliance
Why Digital Maturity Is Essential For Medical Device Manufacturers

Digital maturity is paramount in highly regulated industries, and we’re going to explain why
Why Your Business Should Act Now To Be Compliant With IVDR

Why your business should act now to be compliant with IVDR
Kallik Brings Technology & Industry Expertise To Help Teleflex Deploy A Global Labeling System

How Kallik helped Teleflex revolutionize their labeling
How Managing The Simple Medical Device Label Could Make Or Break Nordic Expansion Ambitions For Man

How effectively managing the simple device label could be key to overcoming the challenges of entering the
Leading Orthopaedics Company Össur Overcomes IFU Management Challenges With Veraciti™ Solution

Overhauls legacy asset management systems and disjointed processes to improve global business operations an
Are you ready for the new Windsor Framework Regulations? FREE guide

